Senolytic drugs have been the most promising near-term anti-aging therapy since the ground-breaking paper by van Deursen of Mayo Clinic published in 2011. The body accumulates senescent cells as we age, damaged cells that send out signal molecules that in turn modify our biochemistry in a toxic, pro-inflammatory direction. Though the number of such cells is small, the damage they do is great. Van Deursen showed that just getting rid of these cells could increase lifespan of mice by ~25%. But he did it with a trick, using genetically engineered mice in which the senescent cells had a built-in self-destruct switch.
After that, the race was on to find chemical agents that would do the same thing without the genetically engineered self-destruct. They must selectively kill senescent cells, while leaving all other cells unharmed. It’s a tall order, because even a little residual toxicity to normal cells can be quite damaging. Before last week, the two best candidates were FOXO4-DRI and a combination of quercetin with dasatinib.
I’ve written in the past (here and here) that senolytic drugs are our best prospect for a near-term lift on the road to anti-aging medicine.
Last week, a large research group affiliated with the original May Clinic team published findings about fisetin, the latest and greatest candidate for a senolitic pill, another flavenoid, very close in structure to quercetin. 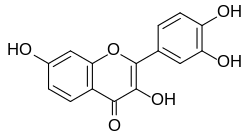
They grew senescent and normal cells in a test tube, then tested 11 different plant-derived chemicals for power to kill the one while leaving the other unharmed. The winner was fisetin.
 (MEF stands for Mouse Embryonic Fybroblast, the cells that were cultured in the screening experiment.)
(MEF stands for Mouse Embryonic Fybroblast, the cells that were cultured in the screening experiment.)
Fisetin is especially interesting because it is cheap, easily available, widely-regarded as safe, but not nearly as well studied as quercetin.
They took the winner, fisetin, and subjected it to a series of tests. They began with in vitro (cell culture) tests and proceeded to in vivo tests with live animals, culminating with an impressive life span assay in mice.
(The runner-up was curcumin, less interesting perhaps only because it has already been extensively studied. The curcumin molecule is unrelated to quercetin or fisetin, and is not a flavenoid. I can’t help but wonder if they had subjected curcumin to the same thorough testing that they reserved for fisetin, how would curcumin have fared?)

curcumin
The paper’s principal findings were:
- Fisetin has lower liver toxicity (at equivalent doses for senolytic benefit) than any of the other senolytics tested so far.
- Fisetin reduces pro-inflammatory signaling in a short course given to mice and in long-term experiments where fisetin was added to the mouse chow.
- Fisetin reduces number of senescent fat cells in a short course given to mice.
- Mice fed fisetin for long periods had much more glutathione than control mice. (Glutathione is one of very few marker molecules that seems to be wholly beneficial.)
- Most impressively, mice fed fisetin late in life lived 10-15% longer than control mice. This represents a 50% increase in the remaining lifespan after the intervention.

What we know and what we’d really like to know
We’d like to know, do humans who take large doses of fisetin live longer? Do they have toxic side-effects? These questions require decades to answer.
Does fisetin reduce age markers in humans, especially methylation age? This is a feasible study, since the test is mature and safety of fisetin is fairly well established for short courses. Perhaps this experiment is being considered; I’ve written to the corresponding authors of the most recent study, in case they haven’t already thought of it. This test would not be definitive because we know that methylation age is not perfectly correlated with biological age; but if positive it would confirm both that fisetin is accomplishing epigenetic rejuvenation and that methylation tests were correctly informing us of this; a negative result would be ambiguous.
Episodic Dosing
It makes sense that senolytics should be taken periodically, not continuously. A high dose can be toxic to existing senescent cells, and then getting out of the way, it can allow normal cells to recover from any damage. This sounds like good theory, but different dosing regimens have not been tested experimentally. In fact, the new paper reports positive results from both high episodic dosing and lower everyday dosing.
The Mayo group had previously tested fisetin, and found it effective in killing some kinds of human senescent cells but not others. In previous tests, fisetin was found to be effective in senescent fat cells (pre-adipocyte, white adipose tissue), and that is where it was primarily tested in the new studies.
Authors’ comments
They note that the episodic treatment and short half-life suggest that the benefits of fisetin come from its senolytic action, rather than other actions as an antioxidant and signal molecule. They emphasize that clearing senescent white blood cells and making room for new, active white blood cells are activities that enhance the benefits of fisetin, since white blood cells contribute to clearing the remaining senescent cells.
Fisetin has previously been shown to have anti-cancer activity and to inhibit inflammatory signals directly. Here is a review of benefits of fisetin from three years ago. Drugage lists just two previous lifespan studies with fisetin, with encouraging results from yeast and fruitflies.
The Bottom Line
If we choose to take fisetin at this stage in the science, we are early adopters, and our main concern ought to be safety. There is little doubt that killing senescent cells will be beneficial. But what is the toxic burden of fisetin, and what dosage can we safely take without risk of damage to normal cells? The current study covers a lot of ground but doesn’t answer this question, apparently because they are convinced that fisetin is quite safe.
Strawberries, apples, grapes, and onions all contain fisetin, but at low levels compared to a senolytic dose. For example, the highest food concentration, 160 ppm, is found in strawberries. A half pound of strawberries yields 36 mg of fisetin. We’re still guessing at the therapeutic dose, based on mouse studies, and the experimental dosage in human trials is about 1,000 to 1,500 mg (based on this clinical trial), the content to 30-40 pounds of strawberries on each of two consecutive days.
In the best cases, fisetin was shown to reduce senescent cell burden by 50% and up to 75% in cell cultures. This is a good start, and encourages us to think we can do better by combining fisetin with other agents, or perhaps with fasting.
Also reported today,
Clearing Senescent Cells From The Brain In Mice Preserves Cognition
It sounds impressive, but I’m not impressed. First, mouse models of Alzheimer’s have been discredited repeatedly. Mice don’t naturally get AD, so they have to be genetically engineered to do so, and the genetically modified mice don’t share the deep causes of human AD. Time and again, treatments have been found effective in the mouse model that fail to translate to humans. Second, the treatment used in the study to kill senescent brain cells also relied on another genetic modification, and would not be applicable to humans.
My guess is that effective senolytic agents for humans will be available within a few years, and that they will decrease risk of all age-related disease, including Alzheimer’s. But this study does little to advance us toward that goal.

Take a look at what Dr Mercola has to say about molecular hydrogen therapy. This is all new to me: https://articles.mercola.com/sites/articles/archive/2018/10/21/molecular-hydrogen-benefits.aspx
Thank you once again for sharing the news, Josh! I’m thinking that another thought in considering episodic rather than everyday dosing of fisetin (or many other supplements) is the former protocol’s greater potential to diminish a homeostatic response, which may in turn diminish these supplements’ effectiveness. Any other thoughts on this?
Yes – I take everything on an irregular schedule, based on this kind of reasoning. I think we’re in uncharted territory.
I hope someone checks the microbiome for the production of H2. I believe that the beneficial microbes are generally anaerobic. Some anaerobes will generate H2 which diffuses quickly and would not be metabolized in an anaerobic environment. To the extent that further research confirms the benefits of H2, the challenge will be to find out how to feed the H2 generators.
It’s true I would be tempted to eat more strawberries if it can rejuvenate me even a bit. But there is a nagging question: What happens when a senescent neuron or a senescent cardiac pacemaker cell dies?
Neurons don’t divide, so they don’t go senescent from short telomere length… but senescent cells don’t perform their normal functions correctly anyway. The senescent fibroblasts that I nursed along for a couple of years blew up to 10X the right size… and they spew pro-inflammatory chemicals.
Cells that capable of senescence, that are killed by a senolytic, are replaced by new cells.
So I’ve read.
Nice coverage Josh. Perhaps in the same ‘natural senolytic’ category, any thoughts on piperlongumine (from Indian Long pepper)? Here’s a link describing one mechanism of action.
https://www.ncbi.nlm.nih.gov/pubmed/29766639
You know, they need to sell everything.
It’s more like you will die sooner.I have found a great excellent senolytic in Russia
Emoxypine (2-ethyl-6-methyl-3-hydroxypyridine), also known as Mexidol
Can you point us towards some evidence that emoxypine is senolytic?
From the same paper http://dx.doi.org/10.18632/aging.101633 a table of ineffective compounds. https://s3-us-west-1.amazonaws.com/paperchase-aging/supplemental_materials/6fAcrvjkSYuFq34BD_sd1.pdf
Also, Azithromycin has been used for years in the treatment of cystic fibrosis. 250 mg daily with great results without any drug resistance. https://doi.org/10.1136/thorax.57.3.212 HIV at 1200 mg weekly. The CF treatment was thought to prevent the onset of the myo-fibroblast phenotype. However, in light of our current data, we suggest instead that myo-fibroblasts are actually senescent cells, which are rapidly and effectively eliminated by Azithromycin.
In case my last post failed because of containing a link. Good coverage of fisetin. Perhaps another ‘natural senolytic’ is piperlongumine (from Indian long pepper). An article on Pubmed describes one mechanism — title is “Oxidation resistance 1 is a novel senolytic target.”
Hey Jeff!!
Does Melatonin act as a better senolytic than fisetin??
lol
why is this question funny?
You should google before you giggle!
Melatonin is better than a senolytic ..it rescues cells from senescence.
Melatonin reverses H2O2-induced premature senescence in …
http://www.ncbi.nlm.nih.gov › Journal List › HHS Author
Melatonin suppresses doxorubicin-induced premature …
https://www.ncbi.nlm.nih.gov/pubmed/22536785
Together, these results reveal that melatonin has an inhibitory effect of melatonin on premature senescence at the cellular level and that melatonin protects A549 cells from DOX-induced senescence. Thus, melatonin might have the therapeutic potential to prevent the side effects of …
Melatonin reverses H2O2‐induced premature senescence in …
onlinelibrary.wiley.com/doi/10.1111/jpi.12250/abstract
Journal of Pineal Research · 2015
Melatonin Rescues Mesenchymal Stem Cells from Senescence …
http://www.biomolther.org/journal/view.html?volume=26&number=4&spage=389&...
Melatonin reverses H2O2-induced senescence in SH-SY5Y …
onlinelibrary.wiley.com/doi/10.1111/jpi.12407/abstract
SH-SY5Y cells were treated with H 2 O 2 to induce the senescent state.
Melatonin regulates PARP1 to control the senescence …
https://reference.medscape.com/medline/abstract/2824753
in fact it dioes it rescues senescent cells and makes them normal try googling it it iis not that difficult just type in the words and hit the search button
lmao
Jeff – you provide excellent information and I enjoy reading your posts.
Thank you for the contribution – I was only joking about your vast over-simplification of Bredesen’s supplement/protocol anti-alzheimer’s regimen (55 items listed) as possibly down to just 1 supplement: melatonin (which is obviously ridiculous).
I think melatonin is beneficial but certainly not the panacea you make it out to be; but if it helps you sell books, carry on.
Fascinating stuff, and something one can easily do at home.
I noticed an interesting exclusion criterion in Dr. Kirkland’s clinical trial, i.e. the concomitant use of proton pump inhibitors. I assume that has to do with the ability of these drugs to inhibit the ABC transporters and increase absorption of polyphenols as described in this interesting paper: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2820202/. I suppose that the extent of the effect is imprecisely known thus far, and would therefore muddy the results. Still, this has the potential to facilitate entry of polyphenols to various tissues, such as the brain, so that more types of senescent cells could potentially be targeted. I hope to encounter more research on this. Does anyone know of any, or have thoughts on the matter?
The second is the
Please ignore that last sentence fragment.
Josh, regarding your idea of testing epigenetic age changes from a senolytic therapy – I think the result would depend on the tissue type. If the cleared cells were then replaced by differentiation from epigenetically younger stem cells then you would expect a positive result. But in some cases cells are turned over very rapidly anyway, and I’m that case I expect an ambiguous result.
Didn’t they show epigenetic age reversal for allogenic stem cell transplant patients?
My major concern about Fisetin is that it acts as a topoisomerase 1 and 2 inhibitor. Various chemotherapy drugs also work via this mechanism and have to found to damage chromosomes and may lead to leukemia, in particular AML.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3961513/
It has also been recently demonstrated that this same mechanism may lead to the tendon ruptures seen with certain antibiotics in the fluoroquinolone class.
The inhibition of topoisomerase 1and 2 is a real concern with Fisetin, especially at high doses.
Paul, this is very interesting. I had no idea fisetin had these powerful properties. As for topoisomerase 2 inhibitors like fluoroquinolones, they should not exert a negative effect on human cell lines (at least in theory), because the affected bacterial enzymes have little homology with mammalian DNA gyrase. The tendon rupture issue remains elusive, but the underlying cause seems to be related to fluoroquinolones toxicity to type 1 collagen.
Cipro impairs mitochondrial energy production and blocks cellular differentiation most likely leading to most of its side effects including tens rupture. This is most likely due to topoisomerase 2 inhibition. Normal doses of Fisetin are of no concern, but the very high dosing may well be a problem. https://www.sciencedaily.com/releases/2018/10/181001101943.htm
Not to mention the association of topoisomerase 1 and 2 inhibition with the development of AML.
Fisetin may be perfectly safe at any dose. We’ll see. But I’m going to wait until we know more.
From https://www.ncbi.nlm.nih.gov/pubmed/20025993
“Fisetin induced neither topo I- nor topo II-DNA complexes, but behaved as a catalytic inhibitor of both enzymes.”
Do fluoroquinolones share the same mechanism of action as fisetin in their topo-1/2 inhibition?
Fluoroquinolones certainly not, they are bacteriostatic, yet fisetin is an antioxidant, nootropic substance.
Paul, EGCG and reseveratrol, which many on this site supplement with, is also on the list. I suppose a daily cup of green tea should still be ok?
Thanks for the heads up, Paul. So now I’m thinking we should wait for the results of the clinical trials before we start messing around with fisetin.
On the other hand, I’m not a very patient person, and, at 52, I’m starting to feel the effects of all these goddamned senescent rabble-rousing freeloaders in all my tissues, so I’ll happily spend some energy trying to find a cheat. Some questions that come to mind:
1) What are the plasma levels at which fisetin has significant senolytic activity, and how do these compare with the levels that mess with topoisomerase?
2) Are the deleterious effects of topoisomerase inhibition limited to rapidly proliferating cells and, if so, what happens to the proliferation rates of these cell types during a fast? Might we be able to mitigate the consequences by taking fisetin during a fast? (I know that leukocytes multiply quickly on refeeding _after_ a fast (see V. Longo), so presumably that would be a bad time to use fisetin.)
3) Would a combination of fisetin and curcumin synergize to produce useful senolytic effects while keeping fisetin concentrations low enough to be safe?
4) Why is it always so goddamned complicated? Where’s my free lunch, dammit?
Hi, Paul:
I have taken fisetin (1500mg) on two consecutive days (End Of december 2018, and End of January 2019)
It happens that about mid February I had developed strong pain in an elbow tendon, and yesterday in a knee tendon.
Is it possible that this may have been caused by fisetin?
rapid osteoblast growth consumes calcium, and hence contractions and strains. The diet change and magnesium and calcium supplementation is necessary. In third month of taking fisetin I got some tendons injuries, but changing diet to include more milk, and adding calcium supplement and increasing magnesium, D3, K2 supplements’ dose seem so far to alleviate symptoms so I can continue with fisetin regime.
Hi, Paul:
Further to my previous question (I.e. if fisetin probably caused tendon pain):
I have noticed that curcumin is a strong inducer of topoisomerases.
Since curcumin is also senolytic we might conclude that if a strong dose of fisetin is taken concurrently with a strong dose of curcumin we would get enhanced senolytic benefit, and a cancelled effect on topoisomerases.
What do you think about that?
curcumin negates the effect of high doses of fisetin, so I would go with one or the other, but not both at the same time.
Ciprofloxacin is a strong topoisomerase 2 inhibitor and is know to cause tendon injuries (among other nasty injuries as well). It seems fair to assume that high doses of fisetin could cause the same issue.
Hi Josh, everyone,
Cellular senescence can be terminated via two distinct methods. one by causing the cell to undergo apoptosis and the other is to regress or resque the cell from senescence by reverting or restoring the health of the cell back to a fully functional cell. I am not sure the researchers have said which mechanism or both are at play here?
I find it interesting that fisetin as researched by Salk institute over the last 10 years is a powerful antioxidant that increases glutathione levels over time. Also they have shown fisetin to have anticancer and antidiabetic effects.
The question is does fisetins antioxidant effects cause senescent cells to be rescued from senescence to the point that they become apoptosis-competent and then eliminate themselves? Or does the antioxidant effects of fisetin even go further with some senescent cell and allow the cell to repair itself enough to become fully functioning again and not undergo apoptosis?
Does fisetin sort of resurrect the ( now considered dead ) free radical theory of aging?
Any thoughts?
Kevin Brown
Curcumin the other very effective senolytic affects topo 1 and 2 as well….
Curcumin Induces High Levels of Topoisomerase I− and II−DNA Complexes in K562 Leukemia Cells
Miguel López-Lázaro†‡, Elaine Willmore†§, Andrew Jobson†⊥, Kathryn L. Gilroy†, Hannah Curtis†, Kay Padget†, and Caroline A. Austin*†
Institute for Cell and Molecular Biosciences, The Medical School, Newcastle University, Newcastle-upon-Tyne, NE2 4HH, United Kingdom, Department of Pharmacology, Faculty of Pharmacy, University of Seville, 41011, Seville, Spain, Northern Institute for Cancer Research, The Medical School, Newcastle University, Newcastle-upon-Tyne, NE2 4HH, United Kingdom, and Laboratory of Molecular Pharmacology, Centre for Cancer Research, National Cancer Institute, National Institutes of Health, Bethesda, Maryland 20892
J. Nat. Prod., 2007, 70 (12), pp 1884–1888
DOI: 10.1021/np070332i
Publication Date (Web): December 13, 2007
Copyright © 2007 The American Chemical Society and American Society of Pharmacognosy
* To whom correspondence should be addressed. Tel: +44 191 222 8864 . Fax: +44 191 222 7424. E-mail: [email protected]., † Institute for Cell and Molecular Biosciences, University of Newcastle., ‡ University of Seville., § Northern Institute for Cancer Research, University of Newcastle., ⊥ National Cancer Institute, Bethesda, MD.
Cite this:J. Nat. Prod. 2007, 70, 12, 1884-1888
Abstract
Abstract Image
Recent data suggest that curcumin, a phytochemical with cancer chemopreventive potential, might be useful in the treatment of several solid and hematological malignancies. DNA topoisomerases (topos) are the target of several drugs commonly used in cancer chemotherapy. These drugs induce topo−DNA complexes with either topo I or topo II; then cellular processing converts these complexes into permanent DNA strand breaks that trigger cell death. Using the TARDIS in vivo assay, this study shows for the first time that curcumin induces topo I and topo II (α and β)−DNA complexes in K562 leukemia cells. A comparative analysis revealed that the levels of these complexes were higher than those induced by several standard topo I and topo II inhibitors at equitoxic doses. Curcumin-induced topo I and topo II−DNA complexes were prevented by the antioxidant N-acetylcysteine; this suggests that, unlike the standard topo inhibitors, reactive oxygen species may mediate the formation of these complexes. Overall, this work shows that curcumin is capable of inducing topo−DNA complexes in cells with both topo I and topo II and increases the evidence suggesting that this dietary agent has potential to be tested in cancer
chemotherapy
and another one
Antagonism between curcumin and the topoisomerase II inhibitor etoposide
A study of DNA damage, cell cycle regulation and death pathways
Ekram M. Saleh, 1 , † * Raafat A El-awady, 2 , 3 , † Nadia A. Eissa, 4 and Wael M. Abdel-Rahman 5
Author information Copyright and License information Disclaimer
This article has been cited by other articles in PMC.
Go to:
Abstract
The use of combinations of chemotherapy and natural products has recently emerged as a new method of cancer therapy, relying on the capacity of certain natural compounds to trigger cell death with low doses of chemotherapeutic agents and few side effects. The current study aims to evaluate the modulatory effects of curcumin (CUR), Nigella sativa (NS) and taurine on etoposide (ETP) cytotoxicity in a panel of cancer cell lines and to identify their underlying mechanisms.
CUR alone showed potent antitumor activity, but surprisingly, its interaction with ETP was antagonistic in four out of five cancer cell lines. Neither taurine nor Nigella sativa affect the sensitivity of cancer cells to ETP. Examination of the DNA damage response machinery (DDR) showed that both ETP and CUR elicited DNA double-strand breaks (DSB) and evoked γ-H2AX foci formation at doses as low as 1 µg/ml. Cell cycle analysis revealed S phase arrest after ETP or CUR application, whereas co-treatment with ETP and CUR led to increased arrest of the cell cycle in S phase (MCF-7 cells) or the accumulation of cells in G2/M phases (HCT116, and HeLa cells). Furthermore, cotreatment with ETP and CUR resulted in modulation of the level of DNA damage induction and repair compared with either agent alone. Electron microscopic examination demonstrated that different modalities of cell death occurred with each treatment. CUR alone induced autophagy, apoptosis and necrosis, whereas ETP alone or in combination with CUR led to apoptosis and necrosis.
Conclusions: Cotreatment with ETP and CUR resulted in an antagonistic interaction. This antagonism is related, in part, to the enhanced arrest of tumor cells in both S and G2/M phases, which prevents the cells from entering M-phase with damaged DNA and, consequently, prevents cell death from occurring. This arrest allows time for the cells to repair DNA damage so that cell cycle -arrested cells can eventually resume cell cycle progression and continue their physiological program.
Curses!
I did a pub med search for reverses and senescence in the title and got thes 18 results>>>
Acquired resistance to BRAFi reverses senescence-like phenotype in mutant BRAF melanoma.
Krayem M, Najem A, Journe F, Morandini R, Sales F, Awada A, Ghanem GE.
Oncotarget. 2018 Aug 7;9(61):31888-31903. doi: 10.18632/oncotarget.25879. eCollection 2018 Aug 7.
PMID: 30159130 Free PMC Article
Similar articles
Select item 30087798
2.
Effects of Oleo Gum Resin of Ferula assa-foetida L. on Senescence in Human Dermal Fibroblasts: – Asafoetida reverses senescence in fibroblasts.
Moghadam FH, Mesbah-Ardakani M, Nasr-Esfahani MH.
J Pharmacopuncture. 2017 Sep;20(3):213-219. doi: 10.3831/KPI.2017.20.025. Epub 2017 Jul 30.
PMID: 30087798 Free PMC Article
Similar articles
Chemical constituents of Asafoetida | Essential Oil
ayurvedicoils.com/tag/chemical-constituents-of-asafoetida
Asafoetida was the most prevalent folk remedy for treating flatulence, colic and cold in children and as a contraceptive medicine. The Arabian pharmacists and scientists like Avicenna, Fakhr al-Din al-Razi and Ibn al-Baitar have talked about the positive effects of Asafoetida on …
Select item 29803744
3.
SIRT1 reverses senescence via enhancing autophagy and attenuates oxidative stress-induced apoptosis through promoting p53 degradation.
Liu T, Ma X, Ouyang T, Chen H, Lin J, Liu J, Xiao Y, Yu J, Huang Y.
Int J Biol Macromol. 2018 Oct 1;117:225-234. doi: 10.1016/j.ijbiomac.2018.05.174. Epub 2018 May 24.
PMID: 29803744
Similar articles
Select item 28774385
4.
Telomerase mRNA Reverses Senescence in Progeria Cells.
Li Y, Zhou G, Bruno IG, Cooke JP.
J Am Coll Cardiol. 2017 Aug 8;70(6):804-805. doi: 10.1016/j.jacc.2017.06.017. No abstract available.
PMID: 28774385 Free Article
Similar articles
Select item 28479002
5.
Inhibition of Bcl-2/xl With ABT-263 Selectively Kills Senescent Type II Pneumocytes and Reverses Persistent Pulmonary Fibrosis Induced by Ionizing Radiation in Mice.
Pan J, Li D, Xu Y, Zhang J, Wang Y, Chen M, Lin S, Huang L, Chung EJ, Citrin DE, Wang Y, Hauer-Jensen M, Zhou D, Meng A.
Int J Radiat Oncol Biol Phys. 2017 Oct 1;99(2):353-361. doi: 10.1016/j.ijrobp.2017.02.216. Epub 2017 Mar 4.
PMID: 28479002
Similar articles
Select item 28295567
6.
Melatonin reverses H2 O2 -induced senescence in SH-SY5Y cells by enhancing autophagy via sirtuin 1 deacetylation of the RelA/p65 subunit of NF-κB.
Nopparat C, Sinjanakhom P, Govitrapong P.
J Pineal Res. 2017 Aug;63(1). doi: 10.1111/jpi.12407. Epub 2017 Apr 12.
PMID: 28295567
Similar articles
Select item 27660040
7.
Dietary rapamycin supplementation reverses age-related vascular dysfunction and oxidative stress, while modulating nutrient-sensing, cell cycle, and senescence pathways.
Lesniewski LA, Seals DR, Walker AE, Henson GD, Blimline MW, Trott DW, Bosshardt GC, LaRocca TJ, Lawson BR, Zigler MC, Donato AJ.
Aging Cell. 2017 Feb;16(1):17-26. doi: 10.1111/acel.12524. Epub 2016 Sep 22.
PMID: 27660040 Free PMC Article
Similar articles
Select item 27350449
8.
NANOG Reverses the Myogenic Differentiation Potential of Senescent Stem Cells by Restoring ACTIN Filamentous Organization and SRF-Dependent Gene Expression.
Mistriotis P, Bajpai VK, Wang X, Rong N, Shahini A, Asmani M, Liang MS, Wang J, Lei P, Liu S, Zhao R, Andreadis ST.
Stem Cells. 2017 Jan;35(1):207-221. doi: 10.1002/stem.2452. Epub 2016 Jul 11.
PMID: 27350449 Free Article
Similar articles
Select item 27048648
9.
Rapamycin reverses the senescent phenotype and improves immunoregulation of mesenchymal stem cells from MRL/lpr mice and systemic lupus erythematosus patients through inhibition of the mTOR signaling pathway.
Gu Z, Tan W, Ji J, Feng G, Meng Y, Da Z, Guo G, Xia Y, Zhu X, Shi G, Cheng C.
Aging (Albany NY). 2016 May;8(5):1102-14. doi: 10.18632/aging.100925.
PMID: 27048648 Free PMC Article
Similar articles
Select item 25975679
10.
Melatonin reverses H2 O2 -induced premature senescence in mesenchymal stem cells via the SIRT1-dependent pathway.
Zhou L, Chen X, Liu T, Gong Y, Chen S, Pan G, Cui W, Luo ZP, Pei M, Yang H, He F.
J Pineal Res. 2015 Sep;59(2):190-205. doi: 10.1111/jpi.12250. Epub 2015 Jul 7.
PMID: 25975679 Free PMC Article
Similar articles
Select item 25725291
11.
Downregulation of PEA-15 reverses G1 arrest, and nuclear and chromatin changes of senescence phenotype via pErk1/2 translocation to nuclei.
Lee YY, Kim HS, Lim IK.
Cell Signal. 2015 Jun;27(6):1102-9. doi: 10.1016/j.cellsig.2015.02.014. Epub 2015 Feb 26.
PMID: 25725291
Similar articles
Select item 23777706
12.
Antisense directed against PS-1 gene decreases brain oxidative markers in aged senescence accelerated mice (SAMP8) and reverses learning and memory impairment: a proteomics study.
Fiorini A, Sultana R, Förster S, Perluigi M, Cenini G, Cini C, Cai J, Klein JB, Farr SA, Niehoff ML, Morley JE, Kumar VB, Butterfield DA.
Free Radic Biol Med. 2013 Dec;65:1-14. doi: 10.1016/j.freeradbiomed.2013.06.017. Epub 2013 Jun 15.
PMID: 23777706 Free PMC Article
Similar articles
Select item 23518600
13.
Repairing DNA damage by XRCC6/KU70 reverses TLR4-deficiency-worsened HCC development via restoring senescence and autophagic flux.
Wang Z, Lin H, Hua F, Hu ZW.
Autophagy. 2013 Jun 1;9(6):925-7. doi: 10.4161/auto.24229. Epub 2013 Mar 21.
PMID: 23518600 Free PMC Article
Similar articles
Select item 22030897
14.
Ginsenoside Rb1 reverses H2O2-induced senescence in human umbilical endothelial cells: involvement of eNOS pathway.
Liu DH, Chen YM, Liu Y, Hao BS, Zhou B, Wu L, Wang M, Chen L, Wu WK, Qian XX.
J Cardiovasc Pharmacol. 2012 Mar;59(3):222-30. doi: 10.1097/FJC.0b013e31823c1d34.
PMID: 22030897
Similar articles
Select item 20351343
15.
Relaxin therapy reverses large artery remodeling and improves arterial compliance in senescent spontaneously hypertensive rats.
Xu Q, Chakravorty A, Bathgate RA, Dart AM, Du XJ.
Hypertension. 2010 May;55(5):1260-6. doi: 10.1161/HYPERTENSIONAHA.109.149369. Epub 2010 Mar 29.
PMID: 20351343
Similar articles
Select item 18202313
16.
Activation of cardiac progenitor cells reverses the failing heart senescent phenotype and prolongs lifespan.
Gonzalez A, Rota M, Nurzynska D, Misao Y, Tillmanns J, Ojaimi C, Padin-Iruegas ME, Müller P, Esposito G, Bearzi C, Vitale S, Dawn B, Sanganalmath SK, Baker M, Hintze TH, Bolli R, Urbanek K, Hosoda T, Anversa P, Kajstura J, Leri A.
Circ Res. 2008 Mar 14;102(5):597-606. doi: 10.1161/CIRCRESAHA.107.165464. Epub 2008 Jan 17.
PMID: 18202313
Similar articles
Select item 15165184
17.
Senescence-induced expression of cytokinin reverses pistil abortion during maize flower development.
Young TE, Geisler-Lee J, Gallie DR.
Plant J. 2004 Jun;38(6):910-22. Erratum in: Plant J. 2014 Apr;78(2):357. Giesler-Lee, Jane [corrected to Geisler-Lee, Jane].
PMID: 15165184 Free Article
Similar articles
Select item 1504830
18.
Testosterone reverses a senescent decline in extrahypothalamic vasopressin mRNA.
Dobie DJ, Miller MA, Raskind MA, Dorsa DM.
Brain Res. 1992 Jun 26;583(1-2):247-52.
PMID: 1504830
Similar articles
I pattern my week with a 4 day protein fasting period followed by a protein feasting period. I try to time my workouts and supplements around this pattern, but I’m still in the process of figuring it out. My 2 strength training weekly workouts bookend each transition, with a HIIT workout during the protein fast. I also plan on a complete fast maybe once a quarter, but have only managed to complete one about 8 months ago. Planning is easy, doing not so much as life happens. My general supplement pattern would be to only take flavonoids and antioxidants on non-workout days or before ed after a workout, and senolytics could be during the complete fasts and possibly during some of the protein fasts. Don’t want too much too often. Maybe once a month?
ok I looked through all 320+ pub med entries that have the word fisetin in the title and cannot present all 141 of them that i saved so ill sned you a truncated list of …just the titles after this brief note….
Fisetin sounds like a wonder drug! it activites Sirt1 , it is a potent antiinflammatory, it protects all sorts of normal cells, it causes all kinds of cancer cells to self destruct…it is an AMPK pathway regulator, it is antidiabetic, anti depressant , it protects tisxsues against various kinds if injury, it is an antifungal, it prevents amyloid bujild up, it helps to treat parkinsons and als…and its activities are enhanced in conjunciton with mealtonin!>>>
Selected items
Items: 141
Select item 12107653
1.
Wogonin and fisetin induction of apoptosis through activation of caspase 3 cascade and alternative expression of p21 protein in hepatocellular carcinoma cells SK-HEP-1.
Chen YC, Shen SC, Lee WR, Lin HY, Ko CH, Shih CM, Yang LL.
Arch Toxicol. 2002 Jun;76(5-6):351-9. Epub 2002 Apr 23.
PMID: 12107653
Similar articles
Select item 12789233
2.
Fisetin, a flavonol, inhibits TH2-type cytokine production by activated human basophils.
Higa S, Hirano T, Kotani M, Matsumoto M, Fujita A, Suemura M, Kawase I, Tanaka T.
J Allergy Clin Immunol. 2003 Jun;111(6):1299-306.
PMID: 12789233
Similar articles
Select item 15153793
3.
Flavonoids such as luteolin, fisetin and apigenin are inhibitors of interleukin-4 and interleukin-13 production by activated human basophils.
Hirano T, Higa S, Arimitsu J, Naka T, Shima Y, Ohshima S, Fujimoto M, Yamadori T, Kawase I, Tanaka T.
Int Arch Allergy Immunol. 2004 Jun;134(2):135-40. Epub 2004 May 17.
PMID: 15153793
Similar articles
Select item 15781213
4.
Chromosomal malsegregation and micronucleus induction in vitro by the DNA topoisomerase II inhibitor fisetin.
Olaharski AJ, Mondrala ST, Eastmond DA.
Mutat Res. 2005 Apr 4;582(1-2):79-86.
PMID: 15781213
Similar articles
Select item 16298737
5.
Neuroprotective properties of the natural phenolic antioxidants curcumin and naringenin but not quercetin and fisetin in a 6-OHDA model of Parkinson’s disease.
Zbarsky V, Datla KP, Parkar S, Rai DK, Aruoma OI, Dexter DT.
Free Radic Res. 2005 Oct;39(10):1119-25.
PMID: 16298737
Similar articles
Select item 16317137
6.
Fisetin inhibits the activities of cyclin-dependent kinases leading to cell cycle arrest in HT-29 human colon cancer cells.
Lu X, Jung Ji, Cho HJ, Lim DY, Lee HS, Chun HS, Kwon DY, Park JH.
J Nutr. 2005 Dec;135(12):2884-90.
PMID: 16317137
Similar articles
Select item 17050681
7.
Flavonoid fisetin promotes ERK-dependent long-term potentiation and enhances memory.
Maher P, Akaishi T, Abe K.
Proc Natl Acad Sci U S A. 2006 Oct 31;103(44):16568-73. Epub 2006 Oct 18.
PMID: 17050681 Free PMC Article
Similar articles
Select item 17079162
8.
Anti-inflammatory activity of fisetin in human mast cells (HMC-1).
Park HH, Lee S, Oh JM, Lee MS, Yoon KH, Park BH, Kim JW, Song H, Kim SH.
Pharmacol Res. 2007 Jan;55(1):31-7. Epub 2006 Oct 10.
PMID: 17079162
Similar articles
Select item 17387141
9.
Fisetin, an inhibitor of cyclin-dependent kinase 6, down-regulates nuclear factor-kappaB-regulated cell proliferation, antiapoptotic and metastatic gene products through the suppression of TAK-1 and receptor-interacting protein-regulated IkappaBalpha kinase activation.
Sung B, Pandey MK, Aggarwal BB.
Mol Pharmacol. 2007 Jun;71(6):1703-14. Epub 2007 Mar 26. Erratum in: Mol Pharmacol. 2012 Jun;81(6):856.
PMID: 17387141 Free Article
Similar articles
Select item 18279803
10.
Suppressive effects of flavonoid fisetin on lipopolysaccharide-induced microglial activation and neurotoxicity.
Zheng LT, Ock J, Kwon BM, Suk K.
Int Immunopharmacol. 2008 Mar;8(3):484-94. doi: 10.1016/j.intimp.2007.12.012. Epub 2008 Jan 16.
PMID: 18279803
Similar articles
Select item 18359761
11.
Fisetin, a novel dietary flavonoid, causes apoptosis and cell cycle arrest in human prostate cancer LNCaP cells.
Khan N, Afaq F, Syed DN, Mukhtar H.
Carcinogenesis. 2008 May;29(5):1049-56. doi: 10.1093/carcin/bgn078. Epub 2008 Mar 20.
PMID: 18359761 Free PMC Article
Similar articles
Select item 18396148
12.
The flavonoid fisetin promotes nerve cell survival from trophic factor withdrawal by enhancement of proteasome activity.
Maher P.
Arch Biochem Biophys. 2008 Aug 15;476(2):139-44. doi: 10.1016/j.abb.2008.03.023. Epub 2008 Mar 26.
PMID: 18396148
Similar articles
Select item 18761054
13.
Structural requirements for the flavonoid fisetin in inhibiting fibril formation of amyloid beta protein.
Akaishi T, Morimoto T, Shibao M, Watanabe S, Sakai-Kato K, Utsunomiya-Tate N, Abe K.
Neurosci Lett. 2008 Oct 31;444(3):280-5. doi: 10.1016/j.neulet.2008.08.052. Epub 2008 Aug 22.
PMID: 18761054
Similar articles
Select item 18922931
14.
A novel dietary flavonoid fisetin inhibits androgen receptor signaling and tumor growth in athymic nude mice.
Khan N, Asim M, Afaq F, Abu Zaid M, Mukhtar H.
Cancer Res. 2008 Oct 15;68(20):8555-63. doi: 10.1158/0008-5472.CAN-08-0240.
PMID: 18922931 Free PMC Article
Similar articles
Select item 18949064
15.
The flavonoid, fisetin, inhibits UV radiation-induced oxidative stress and the activation of NF-kappaB and MAPK signaling in human lens epithelial cells.
Yao K, Zhang L, Zhang Y, Ye P, Zhu N.
Mol Vis. 2008;14:1865-71. Epub 2008 Oct 20.
PMID: 18949064 Free PMC Article
Similar articles
Select item 19037088
16.
A plant flavonoid fisetin induces apoptosis in colon cancer cells by inhibition of COX2 and Wnt/EGFR/NF-kappaB-signaling pathways.
Suh Y, Afaq F, Johnson JJ, Mukhtar H.
Carcinogenesis. 2009 Feb;30(2):300-7. doi: 10.1093/carcin/bgn269. Epub 2008 Nov 26.
PMID: 19037088 Free PMC Article
Similar articles
Select item 19264955
17.
Induction of p53 contributes to apoptosis of HCT-116 human colon cancer cells induced by the dietary compound fisetin.
Lim DY, Park JH.
Am J Physiol Gastrointest Liver Physiol. 2009 May;296(5):G1060-8. doi: 10.1152/ajpgi.90490.2008. Epub 2009 Mar 5.
PMID: 19264955 Free Article
Similar articles
Select item 19395653
18.
Dietary flavonoid fisetin induces a forced exit from mitosis by targeting the mitotic spindle checkpoint.
Salmela AL, Pouwels J, Varis A, Kukkonen AM, Toivonen P, Halonen PK, Perälä M, Kallioniemi O, Gorbsky GJ, Kallio MJ.
Carcinogenesis. 2009 Jun;30(6):1032-40. doi: 10.1093/carcin/bgp101. Epub 2009 Apr 24.
PMID: 19395653 Free PMC Article
Similar articles
Select item 19633975
19.
Antimetastatic potential of fisetin involves inactivation of the PI3K/Akt and JNK signaling pathways with downregulation of MMP-2/9 expressions in prostate cancer PC-3 cells.
Chien CS, Shen KH, Huang JS, Ko SC, Shih YW.
Mol Cell Biochem. 2010 Jan;333(1-2):169-80. doi: 10.1007/s11010-009-0217-z. Epub 2009 Jul 26.
PMID: 19633975
Similar articles
Select item 19670328
20.
Fisetin, a natural flavonoid, targets chemoresistant human pancreatic cancer AsPC-1 cells through DR3-mediated inhibition of NF-kappaB.
Murtaza I, Adhami VM, Hafeez BB, Saleem M, Mukhtar H.
Int J Cancer. 2009 Nov 15;125(10):2465-73. doi: 10.1002/ijc.24628.
PMID: 19670328 Free PMC Article
Similar articles
Select item 19702784
21.
The hydroxyflavone, fisetin, suppresses mast cell activation induced by interaction with activated T cell membranes.
That’s all interesting but there is nothing there to convince me that a very large dose of Fisetin is safe in humans.
Hi Jeff,
Do you agree that many of the effects you listed would be due to its direct or indirect antioxidant effects and not due to it functioning as a signaling agent since mammals do not make it and is not always available in food? Do you agree it seem reasonable that it is its antioxidant effects that is rescuing the senescent cells and either setting them up for apoptosis or for full return to functional competence as I described in my previous reply above?
It seems that the term “senolytic” leaves out the the removal of senescent cells that are truly rescued and return them to functional competence. Isn’t senescent cell rescue better than senescent cell death much of the time? When cells become senescent due to mitochondrial damage it seems that a powerful antioxidant such as fisetin could allow remaining mitochondria to reproduce/fuse and save the cell or if the percentage of good mitochondria is too small at least get enough of them to a level of competence that they can initiate successful apoptosis? Do you agree with this line of thinking or do you feel that fisetin has some direct ability to distinguish and kill senescent cells while leaving normal cells essentially alone?
Thanks Jeff,
Hi Kevin
I pub med searched “fisetin and signaling” in the title and came up with one study only….here is the abstract>>>
Int J Dev Neurosci. 2018 Oct;69:88-96. doi: 10.1016/j.ijdevneu.2018.07.002. Epub 2018 Jul 20.
Fisetin impedes developmental methylmercury neurotoxicity via downregulating apoptotic signalling pathway and upregulating Rho GTPase signalling pathway in hippocampus of F1 generation rats.
Jacob S1, Thangarajan S2.
Author information
Abstract
Methyl mercury is a teratogenic and neurodevelopmental toxicant in the environment. MeHg affects several biological pathways critical for brain development. The present study validated the effect of Fisetin on developmental MeHg exposure induced alterations in mitochondrial apoptotic pathway and Rho GTPase mRNA expressions in hippocampus of F1 generation rats. Pregnant Wistar rats were grouped as Group I : administered with vehicle control, Group II: MeHg (1.5 mg/kg b.w), Group III: MeHg + Fisetin (10 mg/kg b.w), Group IV: MeHg + Fisetin (30 mg/kg b.w), Group V: MeHg + Fisetin (50 mg/kg b.w), Group VI: MeHg + Fisetin (70 mg/kg b.w), Group VII: Fisetin (30 mg/kg b.w) alone. Fisetin reduced mercury accumulation in offspring brain. In hippocampus, Fisetin preserved mitochondrial total thiol status, glutathione antioxidant system, mitochondrial metabolic integrity and respiratory chain activity. Fisetin ameliorated apoptotic signals by preventing Cytochrome c release, down regulating ERK 1/2 and Caspase 3 gene expression. Fisetin also upregulated mRNA expressions of RhoA/Rac1/Cdc42 in hippocampus. Predominant effect of Fisetin was to reduce mercury accumulation in offspring brain there by diminishing the toxic effect of MeHg. Hence we showed that, gestational intake of Fisetin (30 mg/kg b.w.) impedes developmental MeHg neurotoxicity by regulating mitochondrial apoptotic and Rho GTPase signalling molecules and by reducing the mercury accumulation in hippocampus of F1 generation rats.
Here\s one I found with bing>>> Fisetin treatment of preadipocytes reduced the phosphorylation of S6K1 and mTORC1 in a time- and concentration-dependent manner. To further our understanding of how fisetin negatively regulates mTORC1 signaling, we analyzed the phosphorylation of S6K1, mTOR and Akt in fisetin-treated TSC2-knockdown cells.
Here’s one where it protects bones via signalling>>As expected, the RANKL-activated p38 MAPK and JNKs signaling pathways were inhibited by fisetin in “shControl” cells (shCtrl); p38, JNK and c-jun were less phosphorylated in cells cultured in the presence of fisetin . However, fisetin had a lower inhibitory action in “shMKP-1” cells.
So the bottom line is somehow fisetin seems to activate pathways that protect good tissues and kills of bad actors like cancer cells It’s chemical structure looks very much like a DNA base pair and /or steroid hormone ..kind of like vitmain d3 And with all the OH’s sticking off of it it obvioudly has a lot of antioxidant potential..So my guess is it is causing good things by both ways acting as an antioxidant and a pseudo hormone that affects signalling
You can buy 100 mg supplements of Fisetin. Is it worth considering?
Here is an October 15th article by Dr. Mercola on Fisetin.
https://articles.mercola.com/sites/articles/archive/2018/10/15/fisetin-health-benefits.aspx
Also, the wikipedia page on Fisetin shows a great number of sources.
https://en.wikipedia.org/wiki/Fisetin
To prevent ageing one needs to choose a healthy lifestyle in their 20s or 30s or whenever possible, encompassing periodic fasting, eating small meals of predominantly raw, superfoods which include Fisetins and other phytonutrients, pre and probiotics, enzymes, minerals, and vitamins; a practice of daily aerobic movement where the heart is working against one’s weight–pushing against gravity (or the grave); meditation; projects that challenge the memory; quality, no stress relationships; and freedom to choose a life that is happy, peaceful, thrives in nature, grateful, vibrant and meaningful.
Taking a drug such as Fisetin to clear ageing cells to enhance autophagy might help, but is not the answer. The answer is to choose “life” every day, which encompasses the above factors over years, and prevents scrambling for the quick cure later. Unfortunately, most people get caught with too many symptoms, that have been ignored–many of which could have been attended to on the onset with natural therapies. Thus, becoming desperate to be fixed is the norm for a great many of people over 50.
Choosing “life” as defined above is not a terrible idea, but will at most work on the margins. Unfortunately, “death” will choose us by age 120, without regard to the purity of our lifestyle. …And part of that mortal degradation will come from senescent cells, if not sooner, then later. Thus, even the saints of healthy living will eventually stand to benefit from senolytics, no?
I wonder if these senolytic drugs may synergize with short fasts of 3-5 days? I’m thinking here of Valter Longo’s work on the longevity and health benefits of his 5 day fading mimicking diet. Autophagy, apoptosis and stem cells are all upregulated which may make senescent cells more vulnerable?
However, I’ve looked through all of Valter Longo’s papers and don’t see any mention of senescent cells.Surprised that he hasn’t looked at that aspect yet!
Dear Malcolm L. Klien,
I am no scientist and no doctor but I am almost 70 and have been coaching people on fasting programs with colonic irrigation for around 25 years; and I have to say I have seen people in their 40s, 50s, 60s, and 70s shed a great number of old cells as well as toxic material. This is not the fasting mimicking diet. This is the real thing–no food at all, just mineral water, coconut water and mineral broth. After 7 days and some 10 or 20 days, those who come with inflammatory health conditions leave feeling and looking vibrantly healthy. Autophagy, apoptosis, mitophagy, lymphophagy, you see it all. And what a miraculous privlege it is!.
Eliminating senscent cells requires a daily practice, a whole lifestyle, a commitment to eliminate that which contributes to ageing and all that sustains, enhances, uplifts and enlightens your existence. This includes intermittent and periodic fasting and list of other practices–I did mention some of them in my last comment post. . . Hillary
What do you mean ? How can you see that they are shedding a lot of old cells ?
Here is some interesting feedback from people who are self-experimenting with Fisetin.
https://www.fightaging.org/archives/2018/10/animal-data-shows-fisetin-to-be-a-surprisingly-effective-senolytic/
Two points here.
One is that over the past year research is showing a more complex picture of the role of inflammation and senescence. It is now becoming clear that some degree of inflammation and the SASP is necessary for repair and rejuvention, but obviously not to excess, but the goal is careful intervention with senolytics and I don’t feel that we’re at that point yet. Our knowledge is emerging but still very rudimentary.
This is also true about toxicity. We do not know that fisetin is safe at very high doses. The quinolone antibiotics are given generally over a short course and are associated with spontaneous tendon ruptures. This is almost certainly due to topoisomerase inhibition.
I’m not ready to jump in just yet.
The thing is, even though the SASP is required for regeneration, the persistance of these cells shows that this regeneration has not occurred, otherwise they would have been replaced with healthy tissue. So I’m not concerned about losing the transient benefits of senescent cells. They are useful and used rather than apoptosis in those situations when a chunk of tissue needs to be arrested and replaced – but this situation should not persist.
Whether they can be removed safely in the very old is a bigger concern for me. As is the long term effect of forcing your remaining cells to replace them (bringing those cells closer to senescence, on average).
But as there are clinical trials on fisetin in humans going on RIGHT NOW, most of us can surely afford to wait.
I would recommend you research the iodine protocol. Iodine allows apoptosis to occur. People are taking huge doses (many hundreds of times the RDA) and reporting many benefits. At least Google apoptosis and iodine.
I’ve been taking 100mg Iodoral for 5 years now with no adverse effects and no recurrence of my breast cancer.
I don’t know why researchers are not looking at this necessary nutrient.
I do 3-4 5 day dry fasts during the year to increase autophagy and get rid of sentient cells, I wonder if taking scenolytics would help this process.
I also do intermittent fasting and thinking of using senolycs with that as well, anyone have any experience with this?
why dry please? Reference research for dry fasting to increase autophagy? Not to conflict, very interested in your reason for dry fasting?? Thanks for taking a bit of time, if just your viewpoint, still interested in your reason for doing dry. Thanks
Senolytics have become one of the most hopeful near-term interventions for aging. Not sure it’s much use to me as I’m only 34 and have been doing CR since I was 18, but great for older people. Although, I’m much more excited about companies like Unity, who are working on more powerful senolytics. After following life extension for over 15 years, things are finally starting to pick up momentum. We just a win or two for aging therapies (like senolytics) and it’ll encourage even more investment.
I’m producing 98% pure Fisetin from a novel botanical source and presently have 20 kilos per month capacity. Planning to scale this to 1,000 kilos per month in one year. Im personally going to take 500 Mgs. once per week.
Piperlonggumine is much easier due to centuries of use in Ayurveda. I manufactured industrial scale quantities of Phytochemicals for 18 years and have zero concerns about safety. Supply is no problem and I’m taking 300 mgs. of this with the Fisetin while research on dose / efficacy continues.
Luck and logenvity favor the bold.
If, as an early adopter, I had to cycle fisetin, what kind of cycling would you recommend? thank you
I would be guessing. There is no data to support a dosing regimen. For my own purpose, I take a few pills in the morning and evening of fasting days.
– JJM
Thanks!
I took 900 mg of fisetin taken out of capsules and mixed powder with a tsp of mct oil each of all 3 water fasting days for “lunch”. With a little salt added it was delicious. Added a dose of life extension’s new senolytic supplement on day 3.
Quercetin causes double strand breaks. Is it really a senolytic, or just a destructive force, damaging perfectly good cells?
And just to better understand, how often do you fast? I fast one day per week
Why not take the study dose ( being conducted now) ?
1200 mg for each of 2 days, repeat 1 month later.
Do annually.
Or base it on rat data and do 500 mg, for 5 nights, that’s it.
To enhance effect, water fast on dosing days.
Maybe add a dose of Life Extensions new bioavailable Quercetin plus black tea Senolyic product.
So you wouldn’t take it on weekly bases? I was thinking to take it during fasting days, once per week… but I’m not sure, maybe it is dangerous
They should also research substances that are pro-senolytic so we can avoid them. For example are sugar , animal products, fat pro senolytic. Knowing what is harmful senolytically is just as important as knowing what reduces senolytic cells
Is this a new treatment for senescence cells? FDA approved, inexpensive antibiotic when millions are being spent to find a treatment.
One low dose cleaned out 97% of senescence cells without harming the other cells in vitro. Dosing was critical, 50 Ul, no effect 100 Ul great effect. Paper at bottom of page. Open source.
https://medicalxpress.com/news/2018-11-antibiotics-senescent-cells-ageing.html?fbclid=IwAR2-kOvpA_Vcg4PrtwQnetDQw6Uhezs6QWZ-TyM5xIIAPmfNKq7VyBWp6xw
whew!! looking forward to hearing Josh’s thoughts.
and here’s the link to study
https://www.ncbi.nlm.nih.gov/pubmed/30428454
if holds up in ivo a game changer?
worth self-experimentation carefully but geez 97%—very carefully and with an intelligent time break between.
this has gotta be bad reporting? a flawed study” 97% of “all” senescence cells?
The above study quoted by you also included a table of compounds which did not work in the lab on senescence cells.
https://s3-us-west-1.amazonaws.com/paperchase-aging/supplemental_materials/6fAcrvjkSYuFq34BD_sd1.pdf
I would say the real game changer is when they tweek Fisetin to be more bioavailable in vivo – OR – develop a highly lipophilic version of Fisetin that can be absorbed through skin.
Our skin is highly permeable to lipophilic substances as is the cell membrane of all cells. Creating a lipophilic version would mean that it could be applied as an oil all over the body – absorbing like a sponge from all sides – reaching all parts from the outside in – WITHOUT being metabolised by the digestive system.
It would be like taking a bath and arising renewed.
Certainly very encouraging news. One question though. Do we know, how Azithromycin acts on senescent pancreatic beta cells? Although senescent, they still very efficiently excrete insulin in response to glucose.
It’s not difficult to acquire generics of this, where did you find the information re: 100mg, specifically? Have you tried this personally? What impact did you notice? My concern would be short term given it would destroy other bacteria as well, presumably…
IT IS NOT 100 mg if re the antibiotic
IU’s are not mg’s
it is also not 100mg if re fisetin
for senescence cells
as noted tests would correlate to roughly 500mg for fisetin over 5 days HOWEVER poorly bioavailable,
the antibiotic not done on mice yet, closest correlation in vivo math is quite high and may be dangerously high. So definitely not yet till at least mice study done.
100 IU’s, yes; appreciate the clarification. Interesting study, thank you!
I believe the unit symbol is not “IU” for international units but is the Greek letter mu with a capital M, meaning “micromolar” — the concentration in the tissue culture medium. Also, as a clinical internist, I’ve not heard of IUs of azithromycin.
What would an experimental protocol look like? My guess might be 1x per week at 100iu of azithro, all the while ensuring you are taking a quality probiotic and eating fermented foods…Would love to hear everyone’s thoughts on this.
What is the equivalent of 100iu in mg?
I purchased Azithromycin and took a single dose of 500 mg orally. I would not recommend it. Azithromycin is a strong antibiotic and gave me stomach ache and diarrhea for a week.
I also ground up an azithromycin pill and dissolved it in ethanol and PEG 400. I then applied it to my skin, with the idea that it would be absorbed into my skin and kill all the senescent cells. I can report that it had no negative side effects.
To see noticeable positive effects from topical applications will require about 30 days of application because new skin takes about 30 days to appear.
Hm. 500 mg is the normal first-day dose for Azithromycin, with 250 mg/day for four days after that. I’ve taken it many times for upper respiratory infections with very little in the way of side effects. I guess it’s different for different people.
The concentrations required for senolytic effects are ~100 µM. It concentrates very differently in different tissues, but two Z-packs back to back should achieve the necessary dose in many tissues, including lung fibroblasts.
Where did you purchase it? I can only get it with a prescription, and it’s a huge pain to even see a doctor in Canada, let alone to get their permission for a zany experiment like this.
I have noticed that Unity’ first target is ostoarthritis. They inect their senolytic substance into the joint.
Would that make sense for fisetin? In other words, would it be possible to disolve fisetin in approrpiate solvent, and inject to the joint?
How could a dose be calculated, in that case?
Any suggestions will be appreciated
Hello Zisos,
I have dissolved Fisetin in DMSO, and in PEG 400 when making a senolytic skin lotion, but it did not dissolve well in water.
An effective solvent for joints would probably be DMSO at 10% concentration. Though DMSO itself has an analgesic effect which may result in false positive results.
I arrived at the figure of 10% based on this paper –
“Modulating the Structure and Properties of Cell Membranes: The Molecular Mechanism of – Action of Dimethyl Sulfoxide”
Thank you, Craig.
While the analgesic effect might lead to a “false positive” for fisetin, this might not be bad for me. My intention for trying is to relieve pain. So what is important to me, is the combination of DMSO & fisetin.
Also, I would assume that the effect of DMSO will be short term, wheras the effect of fisetin (if it exists), will also be felt longer term.
Since you are knowledgeable on the subject, maybe I can ask another question:
One method of delivery, would probably injection in the joint. That is not easily done at home.
I have heard about ‘iontophoresis’, another method of delivery that in some cases is equivalent to injection. That is easier to try at home.
What is your opinion? Would that be an appropriate method of delivering the DMSO/fisetin to the joint?
It would be best if you found out the protocol that Unity are using. I will contact Judith Campisi, and ask her what solvent mixture is best for joint injection of Fisetin.
I spoke with my university tutor regarding use of DMSO. She is a professor researching flavanoids. She said that any concentration of DMSO over 0.2% is toxic. So, if you do make up a solvent, you should only add 2ml of DMSO per litre.
HI Zisos, I just wanted to let you know that a substance has been discovered that reverses the aging process in mesenchymal bone stem cells to promote osteogenesis and increase bone mass
Ref : Jinlong Sun, Leiguo Ming, Fengqing Shang, Lijuan Shen, Jihua Chen & Yan Jin , (2015) “Apocynin suppression of NADPH oxidase reverses the aging process in mesenchymal stem cells to promote osteogenesis and increase bone mass.”, Scientific Reports volume 5, Article number: 18572
You can copy their protocol of application. The apocynin should greatly improve your osteoarthritis.
If you read up on how this drug can be applied to joints, then you should be able to figure out a way of applying it to your joints.
I will look into it myself, and learn more.
Hi Zisos
“Oxytocin and bone remodelling: Relationships with neuropituitary hormones, bone status and body composition” by Breuil V1, Amri EZ, Panaia-Ferrari P, Testa J, Elabd C, Albert-Sabonnadière C, Roux CH, Ailhaud G, Dani C, Carle GF, Euller-Ziegler L.
(2011)
This paper found a strong correlation between Oxytocin levels and osteoperosis – the correlation coefficient (r) was -0.6 at a probability level of 0.001 – so there is definitely a strong negative correlation.
Oxytocin is readily available on the world market from a reputable supplier. Application is fairly straightforward. You would need to make up a saline solution and add the required concentration of Oxytocin. You would then use a nasal atomiser to inhale the mist. It is absorbed quickly into the blood stream and circulates throughout the body. Oxytocin acts as a signal hormone.
Anyway, I would recommend you read up on Oxytocin and its effect on osteoperosis before doing anything.
The correlation between Oxytocin serum level and bone mineral density was +0.64. Osteoperosis being characterised by low bone mineral density
Oxytocin receptors expressed in osteoblasts are
functional: oxytocin elicits a rapid increase in the concentrations of intracellular calcium and these effects are blocked by the addition of an oxytocin antagonist. Treatment of undifferentiated and differentiated osteoblastic cells with
oxytocin stimulates PGE2 synthesis, which has been shown to increase bone formation. Therefore, oxytocin, or better oxytocin analogs with more favorable bioavailability, may represent potential anabolic bone agents.
“New approaches to the treatment of osteoporosis”
Francisco José López (2018)
I found this paper – which shows that Oxytocin is probably an effective treatment for osteoperosis, since it strongly correlates with bone density.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4200383/
I have purchased Oxytocin from the world market. It comes as a white powder and costs about £200 per gram.
It readily dissolves in 0.9% saline (you can buy this from any chemist ready made in 500ml bottles).
The concentration I use is 13mg of Oxytocin in 100 ml of saline.
I use a nasal spray to administer it. The dose is 1ml per day.
If you want to link with me on Facebook, you can follow my progress – Craig Kooper
As to Alzheimer’s disease, I kinda chuckle at the mouse models, since we don’t yet completely understand what causes it in humans. So we have genetically engineered mice to contract what we THINK is AD, without really knowing what AD is…
fisetin seems to effectively remove senescent cells at doses of 30mg/per 1kg of body weight or higher (effect starts at about 15-20mg/1kg but is very weak at these doses). At doses of 30mg/1kg the effect is visible after just 4 three-daily repetitions of 30x100mg per 100 kg of weight, spaced for every two weeks (ie. after 2 months).
Hello
thanks for the info….
WHat do you eman by “visible” results thanks
not all tissue. But so far the broadest range. Including immune system, which then more efficiently than unaugmented clears it in other types of tissue. Visible means noticeably less wrinkles on hands.
not all types of senescent cells I do not think
165lb, 50 year old, male. Already taking metformin and NMN. No serious health problems as of yet other than a mild hyperlipidemia.
Tried one round of fisetin at the beginning of July 2019
1500mg at night, for four consecutive days
Doctor’s Best brand ordered via Amazon.com
This is more-or-less the Mayo clinic protocol, slightly modified because I bought two bottles, which is about 4 does for my weight and it was easy to just use up the two bottles.
I took the pills with water, no fancy MCT oil or anything like that.
I also experienced a mild heat/tingling fairly rapidly after the first dose. It wasn’t significant enough to keep me from sleeping and it was gone by the morning
The first day I had the clearest sinuses I’ve ever had (I tend to always have slight allergies/hayfever). Loved this. Too bad it didn’t last as I was back to my slightly-stuffy sinus-normal by the morning of the second day
Second morning I was very hungry so I ate a big breakfast. I frequently intermittent fast and so rarely eat before lunch. It’s quite unusual for me to be hungry in the morning.
Third and fourth days no noticeable symptoms or changes from my normal everyday experience.
I’m going to repeat in another month. I had a round of blood tests after the first dosing. I’ll post if there is anything interesting, or not, to report. Of course there are a million variables so it will be hard to tease out any good data, but I will try.
I am on my third round, 2 months apart, using the Mayo protocol for taking fisetin to clear senecent cells. While I have not noticed any ”results”, I am experiencing mild fever. This happened last time as welI, showing up a few hours after 20mg/kg dose. Fever indicates that a heightened immune system is going after something. Let’s hope it’s going after those senecent cells. Of interest, I Quote:
”Exactly how fever works its magic is ill-understood. But research just published in Immunity by Chen Jianfeng of the Institute of Biochemistry and Cell Biology, in Shanghai, sheds light on the matter. Dr Chen’s work shows how febrile temperatures encourage a particular protein to shepherd immune-system cells to sites of infection.”……….could it also Shepherd the immune cells to sites of senecent cells? It is after all, a top notch immune system that keeps senescent cells at bay when we are young.
I’ve done several rounds of fisetin, sometimes with quercetin, sometimes by itself. I’ve tried the standard protocol, and tried seriously ODing on the stuff.
No fevers, though. I did get a headache one time, but it passed. And I haven’t heard of others who take fisetin getting fevers.
So I suspect it’s a coincidence, particularly if you didn’t get a fever the last few times you took it. You may have a bug of some kind. Rest up 🙂
yeah I notied a little fever the next day too after taking 1.2 grams of fisetin the night before
Considering how fiscetin has evolved in such widely diverse plant species as apple, grape, onion, etc, it would be interesting to learn the function(s) it plays in the plants. Also, I cannot be the first to wonder what the health and longevity impacts would be on a transgenic mouse designed to express fiscetin.